Dictionary Definition:
“[Acetals are] organic compounds formed by the condensation of two alcohol molecules with an aldehyde molecule.”
Selected from OxfordDictionaries.com
Functional Definition:
When we start talking about naming molecules it can get very confusing; which is which and how any carbons, and why is there an -al at the end? Let’s start by breaking down the dictionary definition above. Alcohols are any molecule with an -OH group, from big cyclohexanols to methanol. Aldehydes, if you remember from the section on naming from general chemistry are any compound with a -COH. If the acetal is formed by a condensation reaction then you know that a -H is being cleaved to form water with a removed -OH.
The problem that you might find is that a verbal definition for something visual can get even more confusing. Look at the first image in the figure bellow. This format with a central carbon, an attached -H, two -OR groups and one -R group is the configuration for an acetal. Hemiactelas still have an alcohol attached at the primary carbon. You can think of hemi- as half, so that only one of the two alcohols is still attached as an -OH.
Acetal structure: 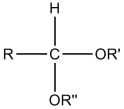
Hemiacetal structure: 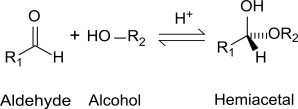
Figure courtesy of Jeff Dahl of Wikipedia Commons