Dictionary Definition:
“[The conjugate acid is] related to the corresponding base by the gain of a proton.”
Selected from OxfordDictionaries.com
Functional Definition:
In a reaction the conjugate acid is the base-H pare or the molecule that was formed by the addition of a proton.
In the reaction mechanism bellow you can see the base represented as -OH and the acid is shown as NH4+. The conjugate acid is the initial -OH with a proton that came off of the acid. The conjugate acid is H-OH (H20).
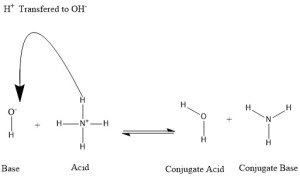
Figure courtesy of Schlenk of Wikipedia Commons