Dictionary Definition:
“A molecule in which a concentration of positive electric charge is separated from a concentration of negative charge.”
Selected from OxfordDictionaries.com
Functional Definition:
A dipole is an electronegative molecule that attracts electrons of partially positive molecules or regions of molecules. The dipole-dipole interactions that are part of inter-molecular forces are the interaction or attraction between the partially negative and partially positive regions of molecules. The easiest example of a dipole-dipole is water and its oxygen and hydrogen attraction. Most molecules contain at least some dipole-dipole interaction.
Bellow is an example of a dipole-dipole interaction of two acetone molecules. Acetone is a common solvent. See the dashed line between the partially negative oxygen and the partially positive central carbon. This line indicates attraction. You can assume that the dipole-dipole interaction is very weak because the oxygen cannot get very close to the carbon which is surrounded by two bulky methyl groups.
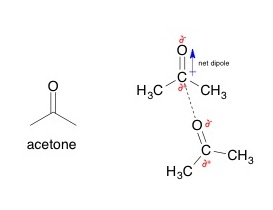
Figure courtesy of AviMole602 of Wikipedia Commons