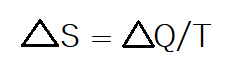Dictionary Definition:
“A thermodynamic quantity representing the unavailability of a system’s thermal energy for conversion into mechanical work, often interpreted as the degree of disorder or randomness in the system.”
Selected from OxfordDictionaries.com
Functional Definition:
Entropy tells us how disordered a system is. Entropy comes from the second law of thermodynamics that tells us that all systems are becoming increasingly disordered over time. This makes sense because ordered states such as ice become disordered or melt as they uptake energy. Entropy is a way to measure the level or amount of disorder currently in a system by dividing the change in heat by the temperature of the system/reaction.