Dictionary Definition:
“A compound or ionic species which can donate an electron pair to an acceptor compound.”
Selected from OxfordDictionaries.com
Functional Definition:
The problem with the Brønsted-Lowry base definition is that it limits what we can consider bases. To broaden how we define as bases we define any molecule that gives an pair of electron to another molecule a Lewis Base. This means that a molecule does not need to take on a proton to be a base.
In the figure bellow the blue boxed molecules are Lewis bases, they are giving a pair of electrons to the molecules they attack or that are at the end of their connecting arrows.
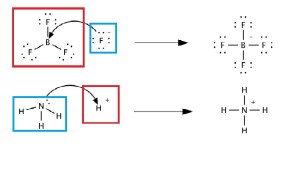
Figure courtesy of Myceteae of Wikipedia Commons