Dictionary Definition of Thermodynamics:
“The branch of physical science that deals with the relations between heat and other forms of energy (such as mechanical, electrical, or chemical energy), and, by extension, of the relationships between all forms of energy.”
Selected from OxfordDictionaries.com
Functional Definition and the Three Laws:
Thermodynamics is how we as chemists, biologists, environmentalists and in general scientists study the movement of energy within systems. There are three laws of thermodynamics that explain these relationships.
- The first law of thermodynamics is calked The Law of Conservation of Energy . The first law says that energy cannot be created or destroyed. In closed systems energy is constant. This means that energy is transferred from one object or system to another rather than being spontaneously formed or destroyed in reactive processes. Think of your body or a thermal reaction like combustion in which energy is produced as heat. This release of heat is the transfer of energy from one system to another (the surrounding environment). Energy can also be transferred as light, in the form of kinetic energy (of movement), gravitational potential, or by radiation. There are a number of ways in which energy can be transferred. This law is expressed my the total change in energy of a system which is equal to the sum of total heat transferred and the amount of work done by the system.
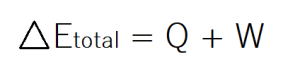
- The second law of thermodynamics saws that the entropy of a system increases over time. As you may know entropy is the disorder of a system. Atomic disorder is considered energetically favorable and systems tend to move toward a state of atomic disorder over time. The best real life example is your room. If you didn’t clean it every weekend it would become incredibly cluttered with dirty clothes, dishes, blankets, and all sorts of day to day things. Your room moves to a state of disorder over time. The equation for entropy says that the total entropy of a system is equal to the heat transferred by/through a system divided by the temperature.
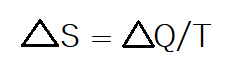
- The third law of thermodynamics says that the entropy of a systems at absolute 0 (zero Kelvin) is 0. At absolute 0 the energetics of a system are at their lowest (called the ground state). It is important to remember that not all systems can be ordered at a realistic ground state (the law typically refers to a crystalline system) and thus there may be residual entropy. You can think of this law as describing an ice cube at absolute zero. The water molecules of the ice cube are not vibrating and are not up-taking any energy from their environment. The ice cube has an entropy of 0.